1.
Introduction
Aging is a mosaic of processes that lead to the decline in physiological
performance and in stress resistance. However, these processes do not always
occur uniformly among different species, strains, individuals or even tissues of
one same individual.
Growing evidence has shown that different individuals of the same species can
achieve the same longevity through different physiological mechanisms
(Vettraino, Buck and Arking, 2001). Environmental factors like diets,
temperature, drugs and physical activity are also involved in the modulation of
aging (Avanesian, Khodayari, Felgner and Jafari, 2010).
Caloric restriction, the most successful experimental intervention in aging, is
known to extend longevity and to improve functional parameters in old age, for
different vertebrate and invertebrate species (You, Sonntag, Leng and Carter,
2007). Caloric restriction may play a role in slowing physiological impairments
in locomotion, sensory functions and cognition (Witte, Fobker, Gellner, Knecht
and Flöel, 2009) as well as in retarding the development of age-related
pathologies (Colman, Beasley, Allison and Weindruch, 2008). With opposing
effects, traits like obesity, as well as fat-rich diets, are often cited as
factors causing longevity shortening and faster onset of age-related pathologies
in a large variety of species (Sun, Seeberger, Alberico, Wang, Wheeler, Schauss
et al., 2010; Bruce-Keller, White, Gupta, Knight, Pistell, Ingram et al., 2010)
. More than a risk factor for age-related pathologies, fat-rich diets are
pointed as a source of perturbation to normal gene expression, being implicated
in common diseases with late-onset phenotypes through interactions between the
epigenome, the genome and the environment (Feinberg, 2007). In the fruit fly
Drosophila, the effects of fat rich diets on aging are poorly studied, although
all available papers show a clear life shortening effect (Driver, Wallis,
Cosopodiotis and Ettershank, 1986; Vermeulen, Van De Zande, Bijlsma, 2006).
However, there are no current studies on age-related functional impairments
influenced by dietary fat in Drosophila. Tests of age-related functional decline
in motor activity of model organisms were used as tools to clarify the
contributions of genetic and environmental influences acting in the lowering of
movement speed (Rhodenizer, Martin, Bhandari, Pletcher and Grotewiel, 2008).The
fruit fly Drosophila melanogaster is one of the most important
experimental models in aging research. This insect has been used in order to
locate the main genes controlling longevity and stress resistance, the
regulation of their expression and the developmental effects of pre-adult
environment in adult lifespan (Arking, 2001). Larval starvation induced by
overcrowded culture media during pre-adult development was studied in extreme
caloric restriction and has been observed to extend adult lifespan and stress
resistance in different strains (Da Cunha and Oliveira, 1996; Rion and Kawecki,
2007; Driver, Wallis, Cosopodiotis and Ettershank, 1986; Vermeulen, Van De
Zande, Bijlsma, 2006). This result suggests that one of the effects of larval
starvation is the activation of developmentally regulated genes with influence
on lifespan.
Among the several phenotypes observed during aging, negative geotaxis is
especially useful to evaluate functional senescence in Drosophila.
Negative geotaxis is an innate escape response in which flies climb the walls of
a container after being tapped to its bottom (Gargano, Martin, Bhandari, and
Grotewiel, 2005). This escape response is known to decline with age in Drosophila
(Rhodenizer, Martin, Bhandari, Pletcher, Grotewiel, 2008). Environmental and
genetic factors acting on the aging process can affect motor activity of the
flies as well (Avanesian, Khodayari, Felgner and Jafari, 2010). Recently, it was
observed that lamotrigine extends Drosophila longevity, although causing
impairments to locomotor activity (Avanesian, Khodayari, Felgner and Jafari,
2010; Burger, Buechel and Kawecki, 2010). Caloric restriction was equally
efficient in order to extend drosophila lifespan, however, it showed no effects
on cognitive aging in this insect or neuronal dysfunction in Drosophila models
of Alzheimer's disease (Kerr, Augustin, Piper, Gandy, Allen, Lovestone et al.,
2009). These data indicate the increasing need of studies addressing different
aspects of functional aging in Drosophila.
Due to the lack of data on functional impairments induced by fat rich diets on
aging in Drosophila, and particularly, on fat consumption during pre-adult
development, the present study aimed to test one wild type and four mutant
strains of Drosophila melanogaster developed under standard and fat-rich
culture media, in order to assess if these different genotypes can be influenced
by pre-adult nutrition conditions in their adult negative geotaxis responses.
2.
Material and methods
2.1.
Drosophila strains
The following Drosophila strains were used:
Wild type Oregon-R controls (Da Cunha and Oliveira, 1996).
ADP60 mutants, showing an obesity phenotype. After 1 week of ad libitum feeding,
the fat body cells of ADP60 flies contain greatly enlarged lipid
droplets compared to controls. In addition, the mutant animals show a twofold
higher accumulation of triglycerides, and a variable, slight reduction of
glycogen levels to 70-80% of normal levels. ADP60adults show increased
resistance to starvation compared to control flies (Häder, Müller, Aguilera,
Eulenberg, Steuernage, Ciossek et al, 2003) IDH ngb2, mutants deficient in
enzyme activity for NADP-dependent isocitric dehydrogenase. This enzyme displays
the same electrophoretic mobility as IdhS. The mutants show only 5% residual
enzyme activity (Burkhart, Montgomery, Langley, 1984).
Men nnc1 flies (deficient for NADP dependent malic enzyme). Enzyme extracted
from MennNC1 homozygotes is kinetically indistinguishable from
wild-type enzyme. It was proposed that MennNC1 is a cis-acting
regulatory mutant. The mutant enzyme has only 3-5% of the normal activity
(Burkhart, Montgomery, Langley, 1984).
CAT n1 individuals (deficient in enzyme activity for catalase). The Catn1
mutation determines a nonfunctioning protein. Decreased viability and longevity
of these acatalasemic flies can be restored by transformation with the wild-type
catalase gene. It suggests that the lethality of these genotypes is due solely
to the lack of catalase (Griswold, Matthews, Bewley, Mahaffey, 1993).
ADP60 mutants were kindly provided by Dr. Ronald Khünlein. Oregon-R strain is
regularly maintained at the Drosophila Lab in the genetics Department at UFRGS.
All the remaining strains were provided by the Bloomington stock center.
2.2.
Culture media and fly handling
The standard culture medium was prepared with 160 g of pre-boiled corn meal, 44
g of molasses, 20 g of soy flour, 36 g of dehydrated yeast, 16 g of agar, 3 g of
nipagin, 1.3 ml of propionic acid, 10 ml of ethanol and 1000 ml of distilled
water.
The fat-rich medium consisted of the standard medium supplemented with the
addition of 200 g of commercially available culinary vegetal hydrogenated fat
(Gordura vegetal Primor, Bunge Alimentos, Brazil).
The flies were maintained at 25 °C and 65% relative humidity, under permanent
light.
Each strain was divided in two groups, one that laid eggs in fat-rich medium,
and one that laid eggs in standard medium. Egg laying time was 3 hours for all
strains. Following eclosion, adults grown in fat-rich and in standard media were
then separately placed in new tubes with standard medium. The fat-rich medium
group was used only for the negative geotaxis experiments and disposed of
thereafter in order to avoid selection for fat tolerance in the strains employed
in the experiments.
2.3.
Negative geotaxis
Negative Geotaxis Assay was performed as follows. Adult males were collected at
the age of 6 days by cold anesthesia (exposure at 0 °C for 3 minutes) and
allowed to recover for at least 18 hours. They were then placed into separate
vials, one tube per strain, 25 individuals in each tube. The individuals
were assayed at the ages of 1 and 3 weeks. Individuals older than 3 weeks were
not available among the group developed in fat rich medium, since fat
consumption reduced maximum lifespan to 23 days (data not shown). The reduced
survival in ages above 3 weeks prevented comparisons to older individuals
cultured under standard conditions. This age limit imposed by fat rich diets has
been observed in other recent studies (Sun, Seeberger, Alberico, Wang, Wheeler,
Schauss, et al., 2010). The individuals to be assayed were placed in the
negative geotaxis vials 30 minutes before the beginning of the test. All tests
were conducted at 25 °C and 65% relative humidity, under permanent light.
A rack containing the negative geotaxis vials was sharply hit 3 times against
the workbench. At the last blow, a photograph was taken using a digital camera
(Canon power shot SD630, 6.0 megapixels, Japan) previously positioned 30 cm away
from the apparatus, with a 4-second delay. The individuals able to climb
to a distance equal or higher than 8 cm in this time interval were counted in
the photos. Each strain was tested 5 times. The flies were given 5 minutes of
rest between each assay.
2.4.
Statistical analysis
The average numbers of climbs of each strain were analyzed by the Repeated
Measures ANOVA test using the SPSS software and Duncan’s multiple pairwise
comparisons between means. Differences between group means were considered
statistically significant if p ≤0.05.
3.
Results
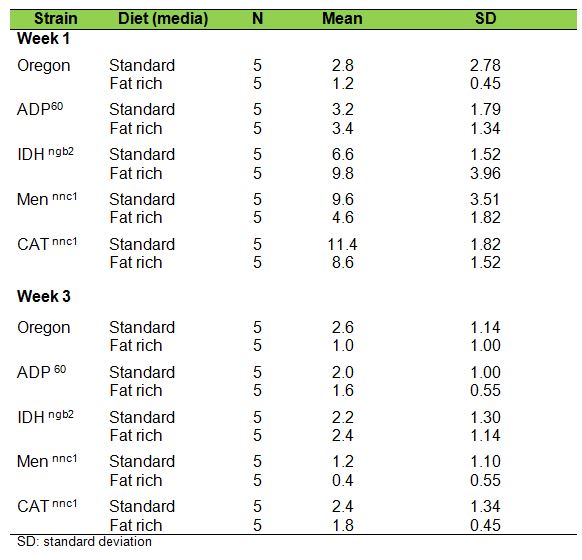
Table 1 presents the observed mean number of climbings in individuals 1 and 3
week old, for the five different samples.
Table
1. Negative geotaxis expressed as climbing means in individuals
one
and three weeks old developed in standard and fat rich media
We observed a significant triple interaction among strain, diet and week (F =
2.661; df= 4, 40 ; p = 0.046) in the within-subjects tests of the repeated
measures ANOVA, meaning that induced diet effects on negative geotaxis were
strain dependent and influenced by age as well. The between-subjects analyses
also revealed interaction effect between strain and diet (F = 5,876; df = 4, 40;
p = 0.003). These results prompted us to compare diets separately by strain and
age of individuals. The results of the pairwise comparisons are presented in
Figure 1 (A and B).
Figure
1. Effect of development under fat-rich media on negative geotaxis of adults
1 and 3 weeks old (A and B respectively): average climbings and standard errors
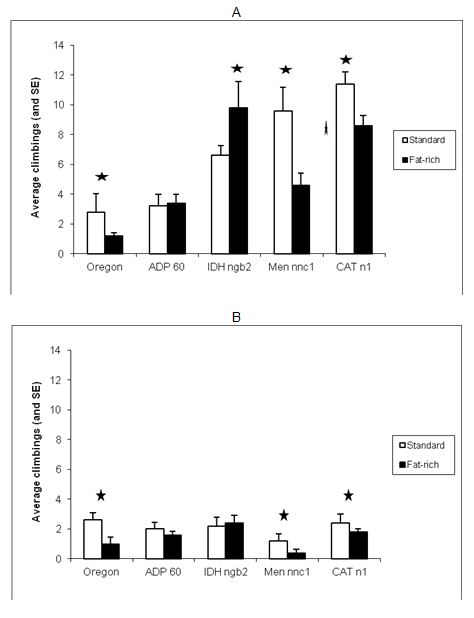
Stars
indicated statistically significant differences between
diets
within strain, using Duncan’s test at the 0.05 level
Among all 1-week-old individuals cultured in standard medium, CATn1 had the
highest negative geotaxis values, followed respectively by Mennnc1, IDHngb2,
ADP60 and Oregon-R. It is interesting to remark that mutants deficient in
catalase, malic enzyme and NADP dependent iscocytric dehydrogenase showed better
performance in negative geotaxis than the Wild type Oregon-R. The same pattern,
however, was not observed in 3-weeks old individuals.
The lowest values in the NG test among all strains, ages and treatments were
those obtained by Mennnc1, 3-weeks old individuals cultured in fat rich media.
This strain’s score in negative geotaxis seemed to be strongly affected both
by age and diet.
In the fat-rich medium, among the 1-week-old individuals of all strains, the
order of successful climbing was IDHngb2> CATn1 > Men nnc1 > ADP60>
Oregon-R, Development in fat-rich medium produced lower
climbing rates both in younger and older CATn1, Men nnc1 and Oregon individuals.
No statistically significant differences between the climbing rates were
observed for ADP60 flies.
Curiously, displaying a radically opposed behavior, IDH ngb2 individuals 1 week
old, grown in fat-rich media, had higher negative geotaxis performances than
their counterparts from standard media and showed no significant differences at
3 weeks old.
3.
Discussion
According to Rhodenizer et al. (2008) the decline in negative geotaxis observed
in Drosophila is age and strain-dependent. In the present paper we
report, for the first time, the effects of development under fat-rich nutrition
conditions in adult performance of five different Drosophila melanogaster
genotypes in the Negative geotaxis test., For three of the five strains studied,
the performances of individuals developed in fat-rich culture medium were poorer
as compared to the performance of their counterparts grown in standard medium. Drosophila
physiology is not identical to mammal physiology. However, at cellular level the
mechanisms induced by dietary manipulation could trigger similar responses in
gene expression (Haigis and Guarente, 2006). There is a reasonable set of
evidences for Drosophila showing that dietary conditions in pre-adult
development can modulate lifespan of adult flies through changes in gene
expression regulation (Arking, Burde, Graves, Hari, Feldman, Zeevi et al.,
2000). In the past decade, different mechanisms were proposed in order to
explain the effects of caloric restriction or high caloric intake on Drosophila
aging. Two of these mechanisms, the dSIR2 mediated histone modifications and
the DNA methylation are among the most studied in Drosophila and in
mammals. Both mechanisms consider caloric restriction as a factor of gene
silencing or, at least, as a gene expression attenuator. A high fat intake is
expected to promote opposing effects through different pathways: a) reducing the
amount of NAD available for the sirtuins could slow down its histone deacetylase
activity (Guarente 2005) and b) a fat-rich diet could disrupt the normal
methylation patterns observed in both development and aging (Burdge, Lillycrop,
Phillips, Slater-Jefferies, Jackson and Hanson, 2009). So far, in Drosophila,
the most plausible candidate of these two mechanisms seems to be the
sirtuins-dependent one, since it is the best documented and the most
experimentally tested. However, the hypothesis of methylation disruption has
been widely investigated in mammals and cannot be entirely ruled out in Drosophila,
because recent evidences showed that intactness of dDnmt2, the only DNA methyl
transferase responsible for DNA methylation in fruit flies, is required for the
maintenance of normal life span. Overexpression of the same gene confers
increased resistance to paraquat in Drosophila melanogaster (Lin, Tang,
Reddy, Shen,2005) Paraquat resistance is often correlated to extended lifespan
phenotypes in Drosophila melanogaster (Arking, 2001).
Despite the unclear nature of the mechanisms acting during the development of
the strains used in the present study, ADP60 mutants — in all ages and media
— exhibited an unusually stable behavior. This gene was identified as an
evolutionarily conserved WD40/tetratricopeptide-repeat-domain protein (Häder,
Müller, Aguilera, Eulenberg, Steuernagel, Ciossek et al., 2003), and was shown
to be a conserved dosage-sensitive anti-obesity gene (Suh, Zeve, McKay, Seo,
Salo, Li et al., 2003). Loss of ADP activity promotes increased fat storage,
which extends the lifespan of mutant flies under starvation conditions.
Curiously, in ADP60 individuals, there were no differences in NG performance in
individuals grown in either the standard or the fat-rich medium, suggesting that
this genotype, in the conditions of the present study, is a factor of greater
influence as compared to the environment (i.e. diets) over the responses to the
NG test.
Since oxidative stress is one of the best known causal factors in motor activity
decline and neurodegenerative diseases, it was expected that CAT n1 individuals
should present the lowest performances, at every experimental condition defined.
Furthermore, considering Malic enzyme and IDH-NADP as important sources of
reducing power, their deficient genotypes (Men nnc1 and IDH ngb2) would be
expected to present lower performances than those shown by the Oregon-R strain.
Remarkably, the highest performance observed for the IDH ngb2 strain was seen in
individuals developed in fat-rich medium. Taking this observation into account,
this set of results suggests that, despite the generally harmful effects of
fat-rich diets on development, the range of responses to these conditions can
vary widely among different genotypes. Additional studies, combining molecular,
biochemical and behavioral assays, are needed in order to identify the
mechanisms underlying the decline of motor activity in different genetic
backgrounds.
Acknowledgements
The authors thank Dr. Ronald Khünlein and the Bloomington Drosophila Stock
Center for providing the mutant strains and to CNPq by grants to Vera Lúcia da
Silva Valente.
Bibliography
-
Arking,
R., Burde, V., Graves, K., Hari, R., Feldman, E., Zeevi, A., Soliman, S.,
Saraiya, A., Buck, S., Vettraino, J. & Sathrasala, K. (2000). Identical
longevity phenotypes are characterized by different patterns of gene
expression and oxidative damage. Exp Gerontol. 35, 353-73.
-
Arking,
R. (2001). Gene expression and regulation in the extended longevity
phenotypes of Drosophila. Ann N Y Acad Sci., 928,157-67.
-
Avanesian,
A., Khodayari, B., Felgner, J. S. & Jafari, M. (2010). Lamotrigine
extends lifespan but compromises health span in Drosophila melanogaster. Biogerontology,
11 (1), 45-52.
-
Bruce-Keller,
A. J., White, C. L., Gupta, S., Knight, A. G., Pistell, P. J., Ingram, D.
K., Morrison, C. D. & Keller, J. N. (2010). NOX activity in brain aging:
Exacerbation by high fat diet. Free Radic Biol Med., 25. Epub ahead
of print.
-
Burdge,
G. C., Lillycrop, K. A., Phillips, E. S., Slater-Jefferies, J. L., Jackson,
A. A. & Hanson, M. A. (2009). Folic acid supplementation during the
juvenile-pubertal period in rats modifies the phenotype and epigenotype
induced by prenatal nutrition. J Nutr., 139, 1054-60.
-
Burger,
J. M., Buechel, S. D. & Kawecki, T. J. (2010). Dietary restriction
affects lifespan but not cognitive aging in Drosophila melanogaster. Aging
Cell, 12. Epub ahead of print.
-
Burkhart,
B. D., Montgomery, E., Langley, C. H. & Voelker, R. A. (1984).
Characterization of Allozyme Null and Low Activity Alleles from Two Natural
Populations of Drosophila melanogaster. Genetics, 107(2), 295-306.
-
Colman,
R. J., Beasley, T. M., Allison, D. B. & Weindruch, R. (2008).
Attenuation of sarcopenia by dietary restriction in rhesus monkeys. J
Gerontol A Biol Sci Med Sci., 63, 556-9.
-
Da
Cunha, G. L. & De Oliveira, A. K. (1996). Citric acid cycle: a
mainstream metabolic pathway influencing life span in Drosophila
melanogaster? Exp Gerontol., 31, 705-15.
-
Driver,
C. J., Wallis, R., Cosopodiotis, G. & Ettershank. G. (1986). Is a fat
metabolite the major diet dependent accelerator of aging? Exp
Gerontology, 21(6), 497-50.
-
Feinberg,
A. P. (2007). Phenotypic plasticity and the epigenetics of human disease. Nature,
447(7143), 433-40.
-
Gargano,
J. W., Martin, I., Bhandari, P. & Grotewiel, M. S. (2005). Rapid
iterative negative geotaxis (RING): a new method for assessing age-related
locomotor decline in Drosophila. Exp Gerontol, 40 (5), 386-95.
-
Griswold,
C. M., Matthews, A. L., Bewley, K. E. & Mahaffey, J. W. (1993).
Molecular characterization and rescue of acatalasemic mutants of Drosophila
melanogaster. Genetics, 134(3), 781-8a.
-
Guarente,
L. (2005). Calorie restriction and SIR2 genes--towards a mechanism. Mech
Ageing Dev., 126, 923-8.
-
Häder,
T., Müller, S., Aguilera, M., Eulenberg, K. G., Steuernagel, A. &
Ciossek, T., et al. (2003). Control of triglyceride storage by a
WD40/TPR-domain protein. EMBO Rep., 4, 511-6.
-
Haigis,
M. C. & Guarente, L. P. (2006). Mammalian sirtuins-emerging roles in
physiology, aging, and calorie restriction. Genes Dev., 20, 2913-21.
-
Kerr,
F., Augustin, H., Piper, M. D., Gandy, C., Allen, M. J., Lovestone, S. &
Partridge L. (2009). Dietary restriction delays aging, but not neuronal
dysfunction, in Drosophila models of Alzheimer's disease. Neurobiol Aging,
5. Epub ahead of print.
-
Lin,
M. J., Tang, L. Y., Reddy, M. N. & Shen, C. K. (2005). DNA
Methyltransferase Gene dDnmt2 and longevity of Drosophila. J Biol Chem.,
280, 861-4.
-
Rhodenizer,
D., Martin, I., Bhandari, P., Pletcher, S. D. & Grotewiel, M. (2008).
Genetic and environmental factors impact age-related impairment of negative
geotaxis in Drosophila by altering age-dependent climbing speed. Exp
Geront., 43, 739-48.
-
Rion,
S. & Kawecki, T. J. (2007). Evolutionary biology of starvation
resistance: what we have learned from Drosophila. J. Evol Biol., 20,
1655-64.
Suh,
J. M., Zeve, D., McKay, R., Seo, J., Salo, Z., Li, R., et al. (2007). Adipose
is a conserved Dosage-Sensitive Antiobesity Gene. Cell Metab., 6(3),
195–207.
Sun,
X., Seeberger, J., Alberico, T., Wang, C., Wheeler, C. T., Schauss, A. G.
& Zou S. (2010). Açai palm fruit (Euterpe oleracea Mart.) pulp improves
survival of flies on a high fat diet. Exp Gerontol., 45(3), 243-51.
Vermeulen,
C. J., Van De Zande, L. & Bijlsma, R. (2006). Developmental and
age-specific effects of selection on divergent virgin life span on fat content
and starvation resistance in Drosophila melanogaster. J Insect Physiol., 52,
910-9.
Vettraino,
J., Buck, S. & Arking, R. (2001). Direct selection for paraquat resistance
in Drosophila results in a different extended longevity phenotype. J
Gerontol A Biol Sci Med Sci., 56, B415-25.
Witte,
A. V., Fobker, M., Gellner, R., Knecht, S. & Flöel, A. (2009). Caloric
restriction improves memory in elderly humans. Proc Natl Acad Sci.,
106, 1255-60.
You,
T., Sonntag, W. E., Leng, X. & Carter, C. S. (2007). Lifelong caloric
restriction and interleukin-6 secretion from adipose tissue: effects on
physical performance decline in aged rats. J Gerontol A Biol Sci Med Sci.,
62, 1082-7.
Another articles in English

|
|
|
EFDeportes.com, Revista
Digital · Año 20 · N° 211 | Buenos Aires,
Diciembre de 2015
Lecturas: Educación Física y Deportes - ISSN 1514-3465 - © 1997-2015 Derechos reservados
|


